|
|
Fats and how they work |
|
Margaret Moss decribes exactly how saturated and unsaturated fats are and how they affect inflammation and clotting. |
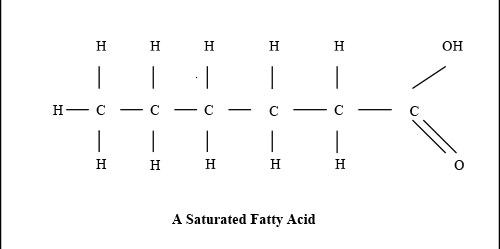
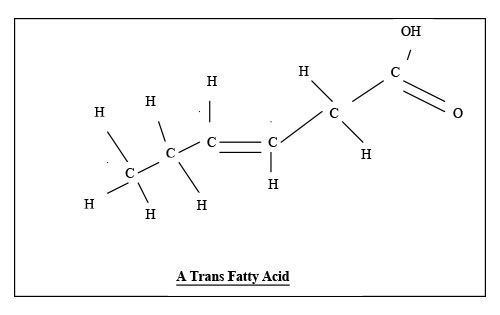
Fats are mixtures of fatty acids. Fatty acids are chains of carbon atoms, C, with hydrogen and oxygen atoms attached to them. There is a hydrogen atom, H, at one end of the chain of carbon atoms. There are two oxygen atoms, O, and a hydrogen one at the other end. Saturated fats are straight. They are saturated with hydrogen, so that is there is nowhere to put another hydrogen atom. Because saturated fats are straight, they fit together neatly, and are solid unless it is hot. Tropical oils like coconut and palm oil are saturated, but can move around the plant in the heat. The lines in the diagram are the bonds that join atoms together. Where there are two lines, it is a double bond. It is like country dancing. Sometimes you hold another dancer’s hand, and sometimes you hold both the other person’s hands.
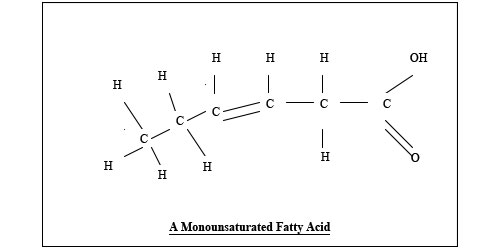
Butyric acid, from animal milk, butter and cheese, has 4 carbon atoms. It is straight, without any bends. So it is 4:0 Butyric, lauric and myristic acids maintain the integrity of the gut, preventing it from leaking large food molecules into the bloodstream, before they have been sufficiently broken down. The end of the molecule with a C and three Hs is known as the methyl or omega end. Omega is the last letter in the Greek alphabet, which is how Omega watches made their claim to be the last word in watches. A small letter omega is ω, and a capital omega is Ω. Monounsaturated fats have one bend, where there is a missing hydrogen atom. The bend has a double bond. Oleic acid, from olives and avocado, is 18:1ω9 (eighteen one omega nine). The nine means the double bond is nine carbons from the omega end of the molecule. The one bend in oleic acid makes olive oil less solid than coconut oil, as the bendy molecules do not fit so neatly together, and so they are more free to move about. Sometimes people use a w, rather than ω, presumably because they look similar, and in the days of typewriters, ω was not available. Others use n, to denote the number of the carbon where the double bond is.
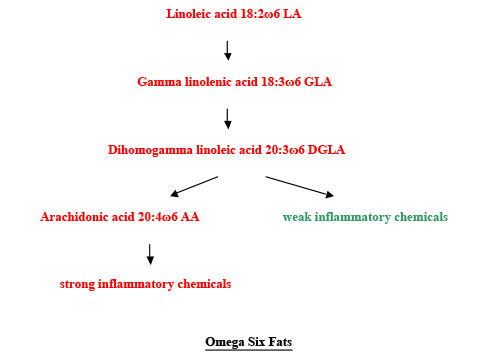
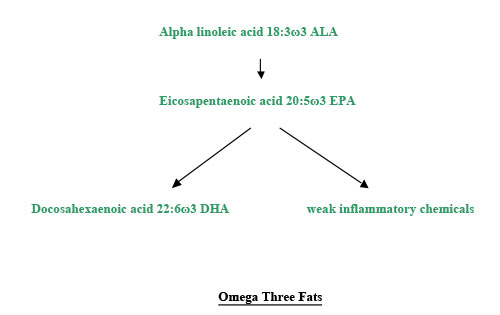
Polyunsaturated fats have more than one double bond, making them more bendy, and more free to move around. They are runny at room temperature. Plants in colder countries have mostly polyunsaturated oils, so they can move round the plant in cold weather. We need two families of polyunsaturated fatty acids to be healthy. One is omega three, which has the first bend three carbons from the omega end. The primary fat in this family is alpha linolenic acid, 18:3ω3, with eighteen carbon atoms, three double bonds, and the first one 3 places from the omega end. Secondary fats can be made from this, just as secondary colours can be made from primary colours. We cannot make 18:3ω3. So it is called an essential fatty acid. It is in flax and hemp seeds and walnuts. The other essential family is omega 6. The primary fatty acid of this family is linoleic acid, 18:2ω6, another fatty acid we cannot make. This is in evening primrose, sunflower, safflower, corn, and sesame oils, and in chicken, turkey and pork. Our bodies have enzymes that convert the two essential fatty acids into others. These enzymes either elongate the fats, by putting more carbon atoms in, or they put more bends in the molecules, making them more unsaturated. These enzymes are called elongases or desaturases. The saturated fat, stearic acid, from meat, is converted by a desaturase to the monounsaturated fat, oleic acid. After the elongases and desaturases have done their work, inflammatory chemicals are made, so that the immune system can fight infection. The omega 6 fats are converted into some mild inflammatory compounds, but also some strong ones. We need these sometimes, but not all the time. Inflammation is involved in many diseases, like coronary heart disease, allergy and rheumatoid arthritis. The omega three fats are converted into mildly inflammatory compounds. Most of the time we would rather have these than the fierce ones. The omega three fats have priority in being processed by the elongases and desaturases. So long as we have plenty of the omega three alpha linolenic acid, we can keep these enzymes busy, so that they don’t produce too many of the fierce inflammatory compounds from the omega six oils, linoleic acid and arachidonic acid.
Some people are short of one of the enzymes involved, delta 6 desaturase. Evening primrose oil provides some GLA, and may be helpful for such people. There is some GLA in blackcurrant seed oil and starflower (or borage) oil, but other constituents in these oils are different from those in evening primrose. I suggest using evening primrose, which has been better researched. Various chemicals are made from the fatty acids. Some of these promote blood clotting. Others reduce clotting. We need a balance between these functions. We don’t want to bleed to death, but we don’t want to die of a heart attack or a clot in the brain either. So we need a balance between omega three and omega six oils. Omega six fats are needed to enhance the action of insulin. The omega three fat, DHA, is needed for the structure of the brain. An increasing number of people try to live without fish or fish oil. Most of them do not realise that they need to find a rich source of omega three fats, like flax or hemp oil. Even if they do use these oils, they are unlikely to have the enzyme capacity to convert enough ALA to EPA and DHA.
The more double bonds, the more fragile an oil is. Runny oils should be kept in the fridge. Only use flax or hemp oils raw. Other runny oils can be cooked briefly, so long as there is water or stock in with them. That way they aren’t heated above the boiling point of water. If you are roasting or frying, use coconut oil, or fat saved from cooking meat, or buy some goose or duck fat. Hydrogenation involves passing hydrogen along a pipe in one direction, and polyunsaturated oil in the other. A metal catalyst is used to incorporate the hydrogen into the oil, to make it less runny, so it will be spreadable or solid. The process of hydrogenation converts polyunsaturated fats in their natural form, called cis, to dangerous trans fats. Instead of having a double bond between two hydrogen atoms on the same side of the carbon chain, these fats are twisted, with one hydrogen atom on one side, and another on the other side. The catalyst may be a combination of nickel and aluminium, which may cause cancer or senility. Some arachidonic acid is in animal fat. This does produce inflammatory chemicals. However, it was safer when the chippies used lard, as saturated fats are not so badly damaged by heat as polyunsaturated fats. Heating these over and over produces trans fats, even if the oils were not hydrogenated.
Fraudulent research led doctors to recommend low fat diets and low saturated fat diets. In fact, fats are essential to life. We need a balance between omega 3 and omega 6 fatty acids, in order to clot when needed, but not when it would be dangerous to do so. We need inflammation to destroy infection, but not perpetual inflammation. We need saturated fats for our intestines, so that they do not become too leaky. What we do not want is hydrogenated or overheated fats. Nutrition and Allergy Clinic |